


The molecular basis of the inner blood-retinal barrier
Breakdown of the inner endothelial blood-retinal barrier (BRB), as occurs in diabetic retinopathy, age-related macular degeneration, retinal vein occlusions, uveitis and other chronic retinal diseases, results
in vasogenic edema and neural tissue damage, causing loss of vision. The central mechanism of
altered BRB function is a change in the permeability characteristics of retinal endothelial cells caused by
elevated levels of growth factors, cytokines, advanced glycation end products, inflammation, hyperglycemia
and loss of pericytes. Subsequently, paracellular but also transcellular transport across the retinal
vascular wall increases via opening of endothelial intercellular junctions and qualitative and quantitative
changes in endothelial caveolar transcellular transport, respectively. Functional changes in pericytes and
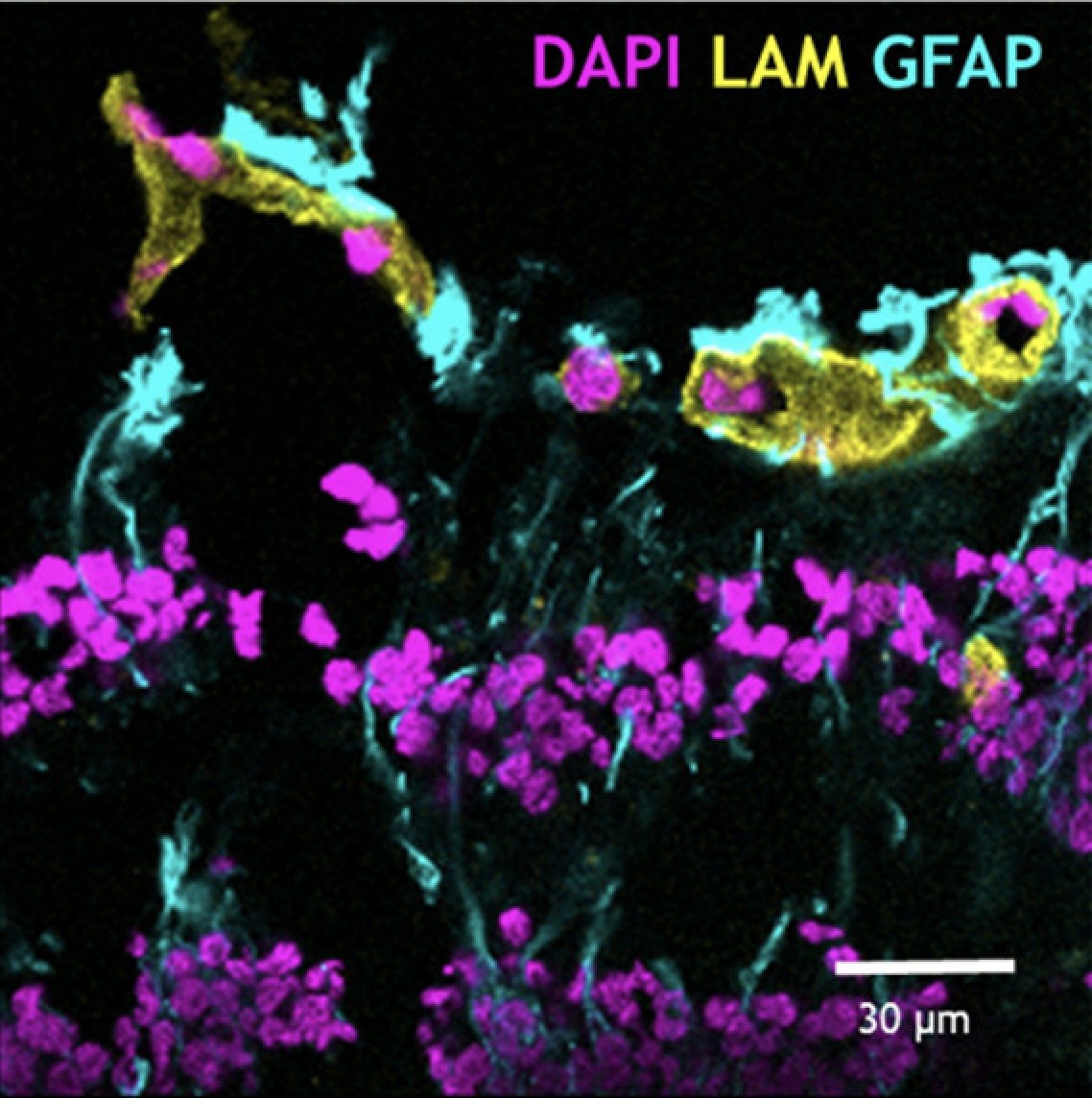
astrocytes, as well as structural changes in the composition of the endothelial glycocalyx and the basal
lamina around BRB endothelium further facilitate BRB leakage.
The inner blood-retinal barrier (BRB) is made up by the neurovascular unit, consisting of endothelial cells,
pericytes and glial cells. The BRB maintains homeostasis of the neural retina, but in pathological eye conditions
the neurovascular unit is often disrupted, causing BRB loss.
Publications:
Several Common Genetic Variations Associate With Functional or Anatomic Effects of Anti-VEGF Treatment in Conditions With Macular Edema.
Invest Ophthalmol Vis Sci. 2025 Jun 2;66(6):2.
Development of Immunostaining Protocols for 3D Visualization of Pericytes in Human Retinal Flatmounts.
JJ Histochem Cytochem. 2025 Mar-Apr;73(3-4):147-170.
Central subfield thickness of diabetic macular edema: Correlation with the aqueous humor proteome.
Mol Vis. 2024 Feb 10;30:17-35.
Differential roles of eNOS in late effects of VEGF-A on hyperpermeability in different types of endothelial cells.
Sci Rep. 2023 Dec 5;13(1):21436.
Disruption of cortical cell type composition and function underlies diabetes-associated cognitive decline.
Diabetologia. 2023 Aug;66(8):1557-1575.
Quantitative Assessment of the Apical and Basolateral Membrane Expression of VEGFR2 and NRP2 in VEGF-A-stimulated Cultured Human Umbilical Vein Endothelial Cells.
Histochem Cytochem (2022) 70:557-569.
Common pathways in dementia and diabetic retinopathy: understanding the mechanisms of diabetes-related cognitive decline.
Trends Endocrinol Metab (2022) 33:50-71.
Expression patterns of endothelial permeability pathways in the development of the blood-retinal barrier in mice.
FASEB J (2019) 33:5320-5333.
The role of plasmalemma vesicle-associated protein in pathological breakdown of blood–brain and blood–retinal barriers: potential novel therapeutic target for cerebral edema and diabetic macular edema.
Fluids and Barriers of the CNS (2018) 15:24.
Molecular basis of the inner blood-retinal barrier and its breakdown in diabetic macular edema and other pathological conditions. Review.
Prog Retin Eye Res (2013) 34:19-48.
Glucocorticoids exert differential effects on the endothelium in an in vitro model of the blood-retinal barrier.
cta Ophthalmol (2019) 97:214-224.
Spatial and temporal recruitment of the neurovascular unit during development of the mouse blood-retinal barrier.
Tissue and Cell (2018) 52:42-50.
TNFα-Induced Disruption of the Blood-Retinal Barrier In Vitro Is Regulated by Intracellular 3',5'-Cyclic Adenosine Monophosphate Levels.
Invest Ophthalmol Vis Sci (2017) 58:3496-3505.
Plasmalemma Vesicle Associated Protein Has a Key Role in Blood-Retinal Barrier Loss.
Am J Pathol (2016) 186:1044-54.
Molecular analysis of blood-retinal barrier loss in the Akimba mouse, a model of advanced diabetic retinopathy.
Exp Eye Res (2014) 122:123-31
A novel co-culture model of the blood-retinal barrier based on primary retinal endothelial cells, pericytes and astrocytes.
Exp Eye Res (2012) 96:181-90.
Altered expression of genes related to blood-retina barrier disruption in streptozotocin-induced diabetes.
Exp Eye Res (2009) 89:4-15.











